Japan approves two world's first iPS cell therapies
On the 6th of March, 2026, the Japan’s Ministry of Health, Labour and Welfare (MHLV) has granted conditional and time-limited approval to two first-of -a- kind regenerative medicines based on human induced pluripotent stem cells (iPSC).
Recent Approvals
The first one to Sumitomo Pharma for Amchepry, an allogenic iPS cell-derived non-frozen dopaminergic neural progenitor cell product for the treatment and improvement of motor symptoms in patients with Parkinson disease who have an inadequate response to existing therapies including levo-dopa derivatives. Dopaminergic neural progenitor cells are cells that have not yet differentiated into dopamine neurons. Patients underwent bilateral brain transplantation of dopamine progenitors at either low-dose (2.1-2.6 million cells) or high-dose (5.3-5.5 million cells) per hemisphere. After cell transplantation, brain imaging (MRI and PET) and neurological assessments were performed at 3, 6, 12, 18 and 24 months. PET studies included 18F-DOPA to assess dopamine synthesis.
The second one to Cuorips Inc, a biotechnology company based in Tokyo for RiHeart a regenerative therapy based on human iPSC-derived cardiomyocyte sheet therapy for the treatment of patients with severe ischemic heart failure. Cuorip’s RiHeart utilises allogenic cardiomyocytes sheets derived from human iPS cells. Once differentiated into heart cells these cells are grown into tiny patches containing up to 100 million cells which are transplanted onto the surface of the heart to support myocardia repair and improve cardiac function.
The Origin and Impact of iPSC Technology
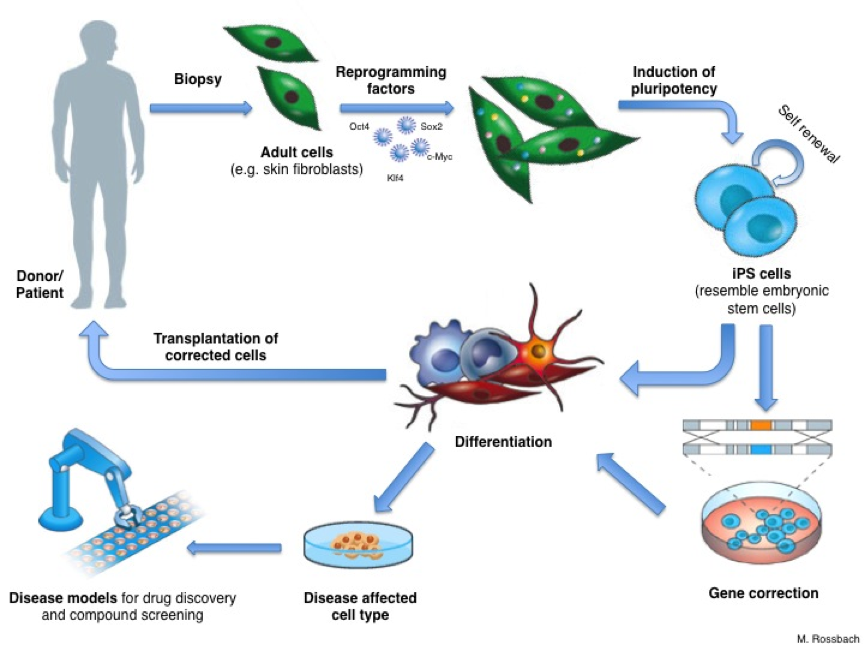
Both Amchepry and RiHeart therapies stem from pioneering technology first described by Takahashi and Yamanaka in 2006 and 2007. Shinya Yamanaka was awarded the Nobel Prize for Physiology or Medicine in 2012 in recognition of this groundbreaking work. Through their research, Takahashi and Yamanaka discovered that mature cells taken from mice and humans could be reprogrammed to become pluripotent stem cells. These induced pluripotent stem cells possess the remarkable ability to develop into any tissue type within the body. (See scheme)

Shinya Yamanaka
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126 , 663–676 (2006).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131 , 861–872 (2007).
Advancements in Cell Biology and Regenerative Medicine
The introduction of iPSC technology has dramatically transformed cell biology research and is now a cornerstone of regenerative medicine. iPS cells are capable of almost unlimited expansion, can be genetically engineered, and may be differentiated into most types of somatic cells. The technology has reached a level of maturity that enables the reliable production of hundreds of millions of high-quality, functional cells suitable for use in human clinical trials and soon in major therapies. In France, Treefrog Therapeutics has developped C-Stem™, a technology platform that integrates GMP compliant encapsulation capable of generating over 1,000 capsules per second. This enables the seeding of up to 10-liter bioreactors, delivering 15 billion iPS cells in a single batch.

Overview of iPSC Clinical Trials
Recent searches on PubMed indicate that there are currently 10 published clinical trials using induced pluripotent stem cells (iPSCs), alongside 22 ongoing studies. Thus far, 115 patients have been treated in these trials. It should be noted that these figures do not include a significant number of ongoing studies in China, which are not reflected in this count.
Key Clinical Applications of iPSC Technology
The most advanced and commonly referenced clinical applications of iPSC-based therapies include:
- Ophthalmology (Retinal diseases such as age-related macular degeneration)
- Parkinson’s Disease
- Cardiology
- Type 1 Diabetes
- Hematology and Oncology
Paris, March 9, 2026
This document has been prepared by Jean-Claude Muller and is provided for information purposes only. The information contained herein has been obtained from sources believed to be reliable but is not warranted to be accurate or complete. The views presented are those of the author at the time of writing and are subject to change. Jean-Claude Muller has no obligation to update these opinions or the information presented.





Comments ()